Bonding
The process of two or more atoms joining together to form a molecule is called bonding. In general, bonding is a chemical change that occurs during chemical reactions. During bonding electrons are involved in "fusing or gluing" two or more atoms together.
There are three general ways in which electrons play a role in bonding.
- They can either be electrostatically moved from the atom of one element to another atom of a second element. This is known as ionic bonding
- They can be shared between two different atoms or they can be shared among the same element of the same atom. This sharing type of bonding is called molecular or covalent.
- Some elements are surrounded by a cloud of free electrons which are shared among all atoms of the same element. This mainly takes place in metallic elements and explains the many properties of metals. This type of bonding is called metallic bonding.
The octet rule of stability:
Why do atoms join other atoms to form new substances called molecules?
The octet rule of stability tells us that all atoms "aspire" to be like noble gases. Noble gases are stable elements because their last orbital (or shell) is completely filled with electrons. This phenomenon does not allow for extra electrons to be placed in the outer shells of these atoms. Therefore these atoms also known as "inert" (non reactive) do not have a "need" to join with other atoms to fill their outer shells.
Apart from the noble gases, most other elements found in the periodic table will have an incomplete outer shell. This means that they will have room to place one or more electrons or to lose one or more electrons in order to achieve stability and look like the noble gases. To to this they will use one of the three methods above. They will "bond" with other atoms and in so doing they will achieve this "octet stability" state. The number of electrons used for bonding is the valence number or the number of electrons found in the outer shell.
Example: Let's start by taking a look at the Sodium Atom's atomic diagram
|
Symbol |
Atomic Number |
Atomic Mass |
# of protons |
# of electrons |
# of neutrons |
|
Na |
11 |
23 |
11 |
11 |
12 |
Atomic Diagram:
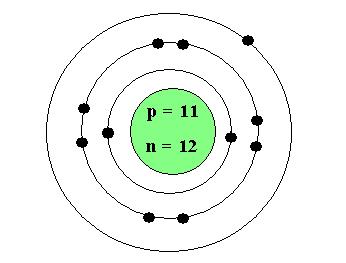
From the above we can see that the sodium atom has one electron in its last shell. Therefore the valence number for sodium is 1.
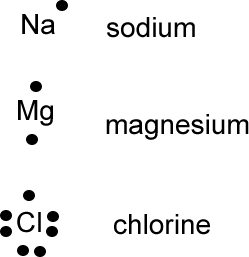
We can use the number of valence electrons to represent the atom using an alternative method to the Bohr diagram. We can simply write the symbol of the atom and a dot for each valence electron around it. This method is known as the Lewis-dot diagram (from the chemist who first thought about using this type of representation).
Examples of Lewis dot diagrams:
1. Ionic Bonding:
For ionic bonding to occur atoms bust become ions first. An ion is an atom which has gained or lost one or more electrons. Atoms in their natural state are neutral. i.e. they have equal number of protons and electrons (positive charges = negative charges). An atom which loses one or more electrons has an overall positive charge (there are more protons than electrons in the atom now). These atoms form positive ions or cations An atom which gains one or more electrons forms negative ions because it will have an overall negative charge now, (there are more electrons than protons in the atom now). Negative ions are also known as anions.
In ionic bonding one or more negative ions will join with one ore more positive ions. The electrons lost by one atom will be "grabbed" by the other atom.
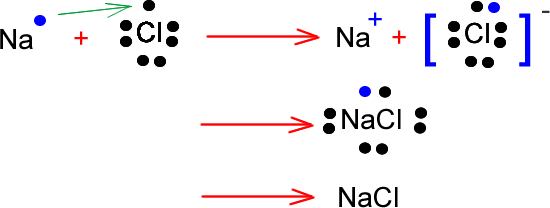
An example of ionic bonding is the one formed between sodium and chlorine when they form Sodium Chloride (NaCl - table salt). Most crystalline substances will form ionic bonds. In these types of substances the positive ions and the negative ions alternate at regular intervals due to the attractive forces acting between them. Therefore they arrange themselves in a regular structure called a lattice. Other examples of ionic bonding are Potassium Fluoride (KF), Magnesium Iodide (MgI2).
This diagram illustrates the formation of sodium chloride. When sodium bonds with chlorine, the sodium atom loses one electron and becomes a positive ion. The chlorine atom gains one electron and becomes a negative ion. The two ions attract each other and join forming a new substance called sodium chloride.
Note that in the process of bonding both atoms become octet stable. They both have their last shell filled with electrons.
Activity: Draw Lewis dot diagrams to explain the formation of the magnesium chloride molecule and the potassium chloride molecule during ionic bonding.
2. Covalent or Molecular Bonding
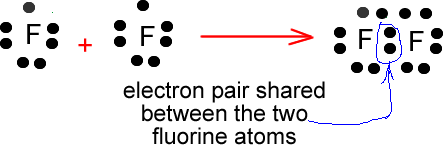
In a covalent bond a pair of electrons is equally shared among two or more atoms. For example hydrogen is very rarely found in nature as a single hydrogen atom. The Lewis structure for hydrogen shows that it has one single electron in its outer shell. Hydrogen "aspires" to be like Helium (the first noble gas). To achieve this state of stability it requires one more electron. One hydrogen atom bonds with another hydrogen atom and thus they will both share two electrons (an electron pair) to form H2(g).
Hydrogen gas therefore is found in nature as a pair of two bonded hydrogen atoms.
This type of a molecule formed by bonding of two identical atoms is known as a diatomic molecule. There are many gases that adopt this type of structure naturally. Some examples are: Oxygen (O2), Fluorine (F2), Nitrogen (N2).
Other examples of molecular bonding are Carbon Dioxide (CO2), Hydrogen Fluoride (HF)
Exercises:
- Draw Lewis dot diagrams to explain the formation of the above molecules
- What type of bond does hydrogen and oxygen form when the water molecule is made?
- Draw the appropriate Lewis dot diagrams and do some research on some of the properties of water such as the anomalous behaviour of water (when water freezes its volume expands instead of decreasing).
- How is this property related to the bonding mechanism found in water?
3. Metallic Bonding:
Metallic bonding occurs in metallic substances. Atoms of metals are held together in this structure by the sharing effect of the electrons amongst all of the atoms. This forms a "sea" or a "cloud" of free electrons that floats around the surface of metals. This cloud of electrons explains many physical properties of metals. In particular, the fact that metals are good conductors can be easily visualized as electric charges are carried from one end of a metal to the other. The fact that metals are shiny can also be explained by this theory. The lustre of metals is in fact due to light (photons) bouncing off the "cloud" of free electrons and creating a shimmering effect.


