Physical Properties of the States of Matter:
|
PROPERTY |
SOLID |
LIQUID |
GAS |
|
shape |
fixed |
same as container |
same as container (indefinite) |
|
volume |
definite |
definite |
fills entire container (indefinite) |
|
ability to flow |
no |
yes |
yes |
|
can be compressed |
very slightly |
very slightly |
yes |
|
volume change with heating |
very small |
small |
large |
Describing Matter
-
Intensive Properties: properties that do not depend on the amount of matter.
Some examples are: colour, odour, density, melting point -
Extensive Properties: properties that do depend on the amount of matter.
Some examples are: mass and volume -
Characteristic Physical Properties:
Properties that are unique for each substance and are used to identify the substance itself.
How to Describe Matter (Qualitative & Quantitative Observations)
1. Physical State: solid, liquid, gas.
2. Colour: green, blue, yellow, black, reddish-brown, etc.
3. Odour: odourless, flowery, spicy, nauseating, etc.
4. Clarity: clear, cloudy, opaque.
5. Luster: shiny, dull.
6. Form: regular (crystalline), irregular (amorphous)
7. Texture: how does it feel? fine, coarse, smooth, waxy, etc.
8. Hardness: can it be scratched easily? scale from 1-10
(e.g. talcum-1, diamond-10)
9. Brittleness: can it break apart or shatter easily? brittle or flexible
10. Malleability: can it be bent and folded into different shapes? malleable or non-malleable
11. Ductility: can it be stretched out into a long wire? ductile or non-ductile
12. Viscosity: can the substance flow? viscous or non-viscous
CHANGES OF STATES OF MATTER
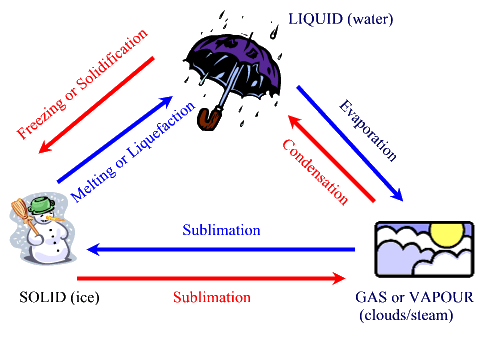
The water cycle, illustrated above, shows the changes of states of water from solid to liquid, to gas.
When a state of matter gains or looses heat it undergoes a change.
A gain in heat is called an endothermic change. A loss in heat is called an exothermic change.
The table below summarizes the six changes of states that matter can undergo.
It also indicates if heat is added or removed for the change to take place.
| Change | From | To | Heat | Examples |
| Sublimation | solid | gas or vapour | added = endothermic | Moth crystals disappear when left in a closet for several days |
| Sublimation | gas or vapour | solid | removed = exothermic | frost forms on a car's windshield |
| Evaporation or vapourization | liquid | gas | added = endothermic | Rain dries up when the sun comes out |
| Melting or Liquefaction | solid | liquid | added = endothermic |
 An ice cube turns into water when left out of the freezer |
| Freezing or Solidification | liquid | solid | removed = exothermic |
 A bottle of water will turn into ice if left in the freezer |
|
Condensation |
gas or vapour | liquid | removed = exothermic | Drops of water form on the mirror when taking a hot shower |


